People often search for the M365 pill after finding an unfamiliar white tablet, receiving a new prescription, or worrying that a loved one may be misusing it. This pill contains hydrocodone and acetaminophen, which means it can be effective for short-term pain relief but also carries serious risks tied to opioid medications, overdose, and addiction. Learning how to identify it, how it works, and when it becomes dangerous can help patients and families make safer decisions.
What is the M365 pill?
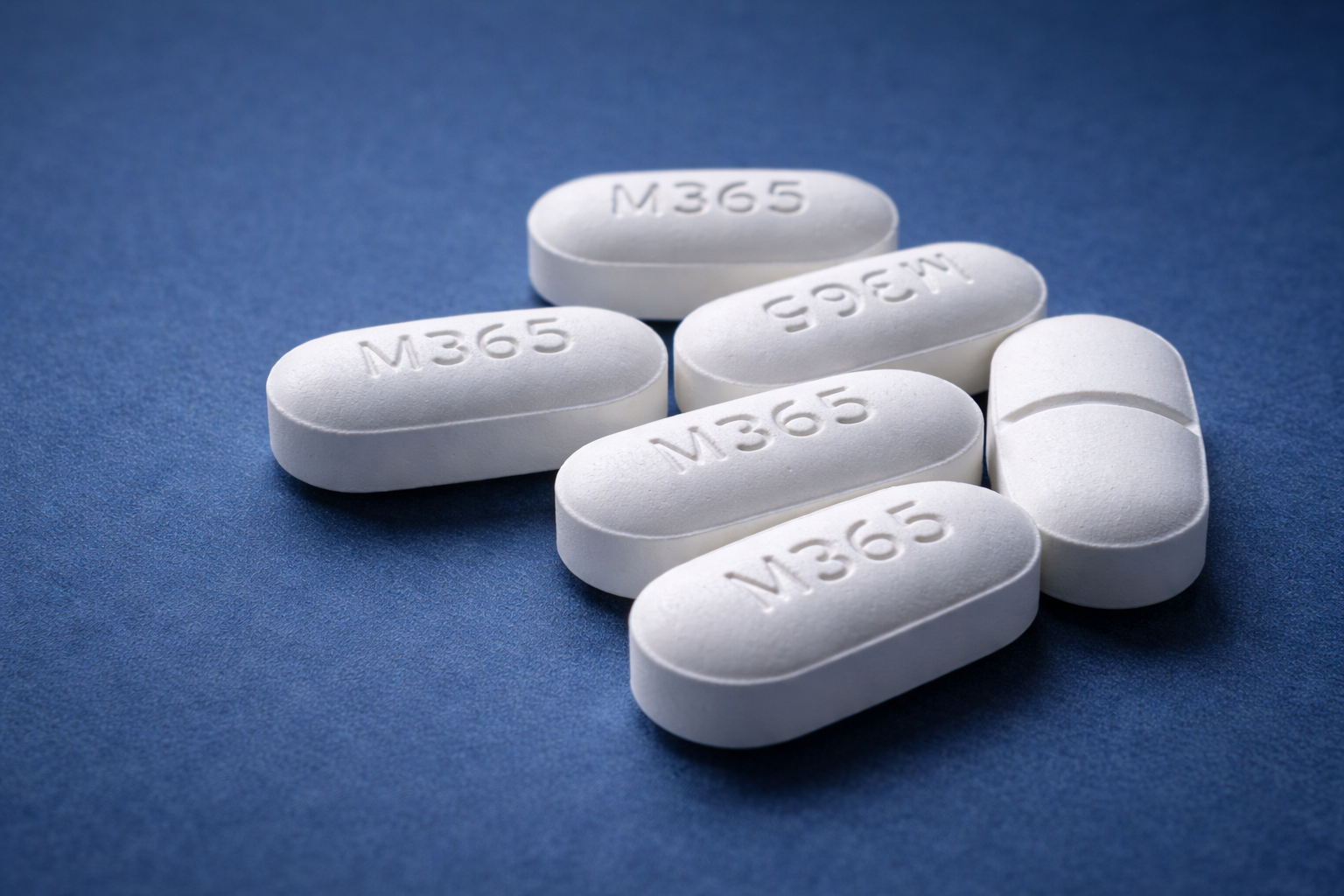
The M365 pill is a prescription combination analgesic manufactured by Mallinckrodt Pharmaceuticals. It contains two active ingredients: hydrocodone bitartrate (5 mg) and acetaminophen (325 mg). You may know the generic name hydrocodone/acetaminophen by brand names like Vicodin, Norco, or Lortab — the M365 is a specific generic formulation within this drug family of opioid medications.
Hydrocodone is a semi-synthetic opioid derived from codeine. Acetaminophen (the active ingredient in Tylenol) is a non-opioid pain reliever included because it enhances hydrocodone’s pain-relieving effect through a separate, non-opioid mechanism. This means the M365 white pill targets pain via two distinct pathways simultaneously. While effective, this combination also carries risks associated with opioid medications, including tolerance, physical dependence, and potential progression to drug abuse when not used as prescribed.
The “M” in the imprint stands for Mallinckrodt, and “365” is the product identifier assigned under FDA regulations. This imprint is what allows pharmacists, emergency physicians, and patients to identify the pill using pill identification databases.
⚠ Controlled substance: prescription required. The M365 white pill is a federally controlled substance (Schedule II under the DEA). It is illegal to possess without a valid prescription.
Pill identifier: how to recognize the M365
If you have found an unidentified white oblong tablet and suspect it may be an M365 white pill, here are its precise physical characteristics:
| Feature | Description |
|---|---|
| Imprint | M365 |
| Shape | Oblong / elliptical (capsule-shaped) |
| Color | White or off-white |
| Score line | Single score (bisect line) on one side |
| Size | Approximately 14 mm × 7 mm |
| Manufacturer | Mallinckrodt Pharmaceuticals |
| NDC | 0406-0365 |
Common search variants: people searching for this pill online use many slightly different terms. “M365 white oblong pill,” “white oval pill m365,” “oblong white pill m365,” “m365 white long pill,” “oval white pill m365,” “white tablet m365,” and “m365 oval white pill” all refer to the same medication. The pill is technically oblong (also called capsule-shaped or caplet-shaped), not a true oval, but both terms are used interchangeably in common usage.
M365 vs. M366 vs. M367
People often confuse the M365 pill with M366 and M367 because they are all white, oblong hydrocodone/acetaminophen tablets from the same product family. The main difference is the strength of hydrocodone. M365 contains 5 mg of hydrocodone and 325 mg of acetaminophen, M366 contains 7.5 mg of hydrocodone and 325 mg of acetaminophen, and M367 contains 10 mg of hydrocodone and 325 mg of acetaminophen. Even though they look similar, they are not interchangeable without a prescriber’s direction, since a higher hydrocodone dose increases the risk of sedation, impairment, and overdose.
⛔ Warning: counterfeit pills are a major overdose risk. A tablet that looks like an M365 pill is not necessarily a legitimate pharmacy-dispensed medication. Counterfeit pills are often made to mimic prescription opioids and may contain illicitly manufactured fentanyl, a powerful synthetic opioid linked to a large share of fatal overdoses in the United States. Pills purchased online, through social media, from a friend, or from any non-pharmacy source carry especially high risk. Even one counterfeit tablet can be dangerous. If a pill is loose, unlabeled, or not in its original pharmacy container, do not take it. Contact a pharmacist or Poison Control at 1-800-222-1222 for guidance on safe handling and disposal.
How the M365 pill works in the body
Hydrocodone: the opioid component
Hydrocodone is a full opioid agonist, meaning it binds directly to mu-opioid receptors (MOR) throughout the central nervous system — primarily in the brain, brainstem, and spinal cord. When it binds to these receptors, it reduces the transmission of pain signals and simultaneously triggers the release of dopamine in the brain’s reward circuitry. This dopamine release is responsible for both the euphoric feeling that makes opioids misuse-prone and the development of tolerance and dependence over time.
Acetaminophen: the non-opioid component
Acetaminophen works through a distinct mechanism. It is believed to inhibit prostaglandin synthesis in the central nervous system, reducing the brain’s sensitivity to pain signals. It also has mild antipyretic (fever-reducing) properties. Critically, acetaminophen does not act on these receptors and has no addiction potential on its own — but in high doses it is directly hepatotoxic (toxic to the liver). The 325 mg dose in M365 is the FDA-mandated maximum per tablet for combination products, reduced from earlier 500 mg formulations precisely because of overdose-related liver failure deaths.
The synergistic effect
The combination of hydrocodone and acetaminophen produces greater pain relief than either drug alone at equivalent doses — a pharmacological synergy. This is why combination products like M365 are often effective for pain that doesn’t respond to acetaminophen or NSAIDs alone.
Medical uses and indications
The M365 pill is FDA-approved for short-term management of moderate to severe pain in adults that is not adequately managed by non-opioid medications alone. Common clinical indications include:
- Post-surgical pain. Severe pain following orthopedic procedures, abdominal surgeries, or other operations where opioid-level analgesia is warranted short-term.
- Dental pain. Severe toothache or post-extraction pain that OTC analgesics cannot control.
- Acute injury pain. Fractures, sprains, or soft-tissue injuries requiring short-term opioid management.
- Cancer pain. As part of a stepped analgesic ladder when milder options are insufficient.
- Chronic pain (limited use). Some providers prescribe it for conditions like severe osteoarthritis, though current guidelines strongly discourage long-term opioid therapy for chronic non-cancer pain due to dependence risks.
ℹ Note. Despite acetaminophen being available over the counter, the M365 pill requires a written prescription from a licensed prescriber. Online pharmacies offering hydrocodone combinations without a prescription are operating illegally.
Dosage information and guidelines
The M365 pill specifically contains 5 mg hydrocodone / 325 mg acetaminophen — the lowest standard hydrocodone dose in the product family.
Hydrocodone/Acetaminophen tablet strengths comparison
| Imprint | Hydrocodone | Acetaminophen | Relative opioid dose |
|---|---|---|---|
| M365 | 5 mg | 325 mg | Lowest |
| M366 | 7.5 mg | 325 mg | Moderate |
| M367 | 10 mg | 325 mg | Higher |
| Various (older) | 5–10 mg | 500 mg | Higher APAP risk |
Standard adult dosage
The typical prescribed dose for adults is 1 to 2 tablets (5-10 mg hydrocodone) every 4 to 6 hours as needed for pain management. The total daily dose must not exceed 8 tablets (40 mg hydrocodone / 2,600 mg acetaminophen) within a 24-hour period without specific medical authorization. The FDA recommends using the minimum effective dose for the shortest duration necessary.
Acetaminophen daily limit
The FDA recommends a maximum of 4,000 mg of acetaminophen per day for healthy adults, and 2,000 mg per day for those with liver disease, heavy alcohol use, or malnourishment. When taking M365, patients must factor in acetaminophen from all sources — including OTC products like Tylenol, NyQuil, or Excedrin — to avoid inadvertent overdose and acute liver failure.
Why hidden acetaminophen is a common problem
One of the most common safety issues with the M365 pill is accidental acetaminophen overuse. A person may take M365 as prescribed, then also use products such as Tylenol, NyQuil, DayQuil, Excedrin, or store-brand cold and flu medicine without realizing they contain the same ingredient. This can raise the total daily acetaminophen dose high enough to cause serious liver injury. Reading labels carefully is essential any time hydrocodone/acetaminophen is being used.
Special populations
- Elderly patients. More sensitive to opioid effects; often require lower starting doses and more careful monitoring for respiratory depression and confusion.
- Renal/hepatic impairment. Both hydrocodone and acetaminophen are metabolized in the liver and cleared via the kidneys; impaired function increases risk of drug accumulation and toxicity.
- Pregnancy. Opioid use during pregnancy is associated with neonatal withdrawal syndrome (NOWS). Use only if the benefit clearly outweighs the risk, under close obstetric supervision.
- Pediatric patients. Not approved for children under 18 in this formulation without specific clinical guidance.
How to take M365 safely
M365 should only be taken exactly as prescribed. Patients should never take larger doses, take it more often than directed, or use it for reasons other than pain relief. It is important to check all over-the-counter medications for acetaminophen, since many cold, flu, sleep, and pain products contain it and can push the total daily dose into a dangerous range. Alcohol, benzodiazepines, and other sedating substances should be avoided unless a prescriber specifically approves them. Patients should not share this medication, save it for future pain without guidance, or stop it abruptly after regular use without speaking to a healthcare provider. For people who are prescribed opioid medications, asking about naloxone is another reasonable safety step.

Common side effects
The following side effects occur in a significant proportion of patients taking the M365 pill, particularly during the first days of therapy or with dose increases. Most are dose-dependent and diminish as the body adjusts.
| Side effect | Notes |
|---|---|
| Nausea / vomiting | Very common; often improves when taken with food |
| Constipation | Universal with opioids; may persist throughout use |
| Drowsiness / sedation | Impairs driving; do not operate machinery |
| Dizziness / lightheadedness | Risk of falls, especially in elderly patients |
| Headache | Common, usually mild |
| Dry mouth | Stay well-hydrated |
| Itching / pruritus | Caused by opioid-mediated histamine release |
| Mood changes / euphoria | Underlies addiction potential |
Contact your healthcare provider if any side effect is persistent, worsening, or interfering with daily activities.
When to call a doctor and when to call 911
Mild nausea, constipation, drowsiness, and dizziness can occur with prescribed opioid use, but symptoms should still be monitored closely. A healthcare provider should be contacted if side effects are getting worse, if sedation becomes more pronounced, if constipation becomes severe, if a rash develops, or if the medication no longer seems to be used as directed. Emergency help is needed right away if breathing becomes slow or shallow, the person cannot be awakened, the lips turn blue or gray, there is choking or gurgling, or the person collapses. These are signs of a possible overdose and should be treated as a medical emergency.
Serious risks and warnings
The FDA has issued its strongest warning — a Boxed Warning — for hydrocodone/acetaminophen products. These are the most clinically important risks:
- Respiratory depression. Slowed or stopped breathing; the #1 cause of opioid overdose death
- Hepatotoxicity. Liver failure from acetaminophen excess; can be fatal
- Physical dependence. Can develop within weeks of regular use
- Addiction (Opioid Use Disorder). 8–12% of chronic-pain patients develop OUD
- Neonatal opioid withdrawal syndrome. Severe withdrawal in newborns exposed in utero
- Serotonin syndrome. When combined with serotonergic drugs (SSRIs, SNRIs, MAOIs)
- Anaphylaxis. Rare but life-threatening allergic reaction
- Adrenal insufficiency. With prolonged opioid use; presents as fatigue and low blood pressure
Seek emergency medical care if any of these develop acutely.
Who is at higher risk from M365?
Some people face a higher risk of serious complications from the M365 pill. This includes older adults, people with liver disease, people with kidney impairment, people with sleep apnea or other breathing disorders, and anyone taking other central nervous system depressants. The risk is also higher in people with a personal or family history of substance use disorder, those with certain mental health conditions, and those who use alcohol heavily. In these situations, even standard doses may require closer monitoring and greater caution.
Drug interactions: what not to mix with M365
Always disclose all medications — including OTC drugs, supplements, and herbal products — to your prescriber before starting M365.
| Substance / drug class | Risk level | Effect |
|---|---|---|
| Alcohol | HIGH | Additive CNS/depression; significantly increases overdose risk |
| Benzodiazepines (Xanax, Valium, Klonopin) | HIGH | Profound sedation, breathing failure, coma, death — FDA Black Box Warning |
| Other opioids | HIGH | Additive opioid effects; escalated overdose risk |
| MAO Inhibitors (phenelzine, selegiline) | HIGH | Serotonin syndrome, severe hypertension — do not use within 14 days of MAOI |
| Muscle relaxants (cyclobenzaprine) | HIGH | Enhanced CNS depression |
| SSRIs / SNRIs (fluoxetine, venlafaxine) | MODERATE | Potential serotonin syndrome; reduces hydrocodone metabolism via CYP2D6 inhibition |
| Antihistamines (diphenhydramine/Benadryl) | MODERATE | Enhanced sedation |
| Rifampin / St. John’s Wort | MODERATE | CYP3A4 inducers — reduce hydrocodone blood levels, reducing efficacy |
| Other acetaminophen products | MODERATE | Risk of exceeding safe daily APAP limit → liver damage |
| Warfarin | LOWER | Acetaminophen may enhance anticoagulant effect at high doses; monitor INR |
Overdose: signs, symptoms, and emergency response
An M365 overdose is a life-threatening medical emergency. Two distinct overdose syndromes can occur: opioid toxidrome (from hydrocodone excess) and acetaminophen hepatotoxicity (from APAP excess — which can be delayed by hours to days).
Signs of opioid overdose (call 911 immediately)
- Slow, shallow, or stopped breathing
- Pinpoint (very small) pupils
- Unconscious or unresponsive
- Blue or grayish lips, fingernails
- Choking or gurgling sounds
- Limp body
- Cold, clammy, pale skin
- Extremely slow heart rate
If an overdose is suspected, call 911 immediately and administer naloxone (Narcan) if available. Narcan is now available without a prescription at most pharmacies.
Naloxone (Narcan): the opioid reversal agent
Naloxone rapidly reverses opioid overdose by blocking opioid receptors. It is available as a nasal spray (Narcan, Kloxxado) and as an injectable. The FDA approved over-the-counter naloxone in 2023. Households of people prescribed opioids are encouraged to keep naloxone on hand. Note that naloxone does not reverse acetaminophen toxicity — if a massive acetaminophen dose has been taken, emergency room treatment with N-acetylcysteine (NAC) is required.
Addiction, dependence, and opioid use disorder
The distinction between physical dependence and addiction is clinically important:
- Physical dependence means the body has adapted to the drug’s presence and will produce withdrawal symptoms upon abrupt cessation. It can occur in anyone who takes opioids regularly for several weeks — it is not the same as addiction.
- Opioid use disorder (OUD) / addiction is a chronic brain disease characterized by compulsive drug-seeking behavior despite harmful consequences. It involves changes in the brain’s reward, motivation, and memory circuitry that persist long after the drug is stopped.
According to the National Library of Medicine, an estimated 21–29% of patients prescribed opioids for chronic pain misuse them, and 8–12% develop an opioid use disorder. The risk is higher in individuals with a personal or family history of substance use disorder, a history of mental health conditions (depression, anxiety, PTSD), younger age, and higher doses or longer duration of use.
⚠ Early warning signs of M365 pill misuse. Taking more than prescribed, running out of pills before the next refill, seeking prescriptions from multiple providers (“doctor shopping”), using the medication for mood regulation rather than pain, feeling unable to function without it, and withdrawing from normal activities are all warning signs that professional evaluation is warranted.

Opioid withdrawal: what to expect
When M365 is stopped abruptly after regular use, the body responds with a withdrawal syndrome. This is not life-threatening in healthy adults, but it is intensely uncomfortable and is a primary driver of relapse. Symptoms typically begin 8–24 hours after the last dose and peak around 36–72 hours.
| Timeframe | Withdrawal symptoms | Severity |
|---|---|---|
| 8–24 hours | Anxiety, restlessness, yawning, watery eyes, runny nose, sweating | Mild–Moderate |
| 24–48 hours | Muscle aches, insomnia, agitation, goosebumps, dilated pupils | Moderate–Severe |
| 48–72 hours | Nausea, vomiting, diarrhea, abdominal cramping, rapid heartbeat, hypertension (peak intensity) | Severe |
| 5–7 days | Physical symptoms begin to resolve; psychological symptoms (cravings, mood instability) persist | Moderate |
| Weeks–months | Post-acute withdrawal syndrome (PAWS): persistent low mood, sleep disruption, cravings | Mild but prolonged |
Medical management of withdrawal can significantly reduce discomfort and improve outcomes. Medications such as buprenorphine, methadone, lofexidine (Lucemyra), and clonidine are used in a detox center Indiana to ease the process.
Safe storage and disposal
Storage guidelines
- Store at room temperature: 68°F–77°F (20°C–25°C), away from heat and moisture
- Keep in the original pharmacy-dispensed container with child-resistant closure
- Store in a locked medication cabinet — not a bathroom medicine cabinet — to prevent diversion or accidental pediatric ingestion
- Keep a count of your pills and note any discrepancy that could indicate theft
- Never share prescription opioids with anyone else — this is illegal and potentially fatal to the recipient
Disposal
The FDA’s preferred disposal method is drug take-back programs — offered at many pharmacies, DEA-registered collectors, and periodic National Take-Back Day events. If no take-back program is available, certain opioids — including hydrocodone combinations — are on the FDA’s flush list, meaning they may be disposed of by flushing. Check the FDA’s flush list or call 1-800-222-1222 (Poison Control) for current guidance.
Safer alternatives for pain management
For many types of pain, especially chronic or recurring pain, there are effective non-opioid or lower-risk alternatives:
- NSAIDs (ibuprofen, naproxen). Anti-inflammatory action; effective for musculoskeletal and dental pain. GI and cardiovascular considerations with long-term use.
- Physical therapy. Builds strength, improves mobility, and addresses root causes of chronic pain — especially effective for back and joint pain.
- Topical analgesics. Diclofenac gel, lidocaine patches, and capsaicin cream target local pain with minimal systemic absorption.
- SNRIs / TCAs. Duloxetine and certain tricyclics have FDA approval or strong evidence for neuropathic pain and fibromyalgia.
- CBT for pain. Cognitive behavioral therapy can help people manage pain-related distress, reduce fear around symptoms, and improve daily functioning, especially in chronic pain conditions.
- Interventional procedures. Nerve blocks, epidural steroid injections, and radiofrequency ablation for specific pain conditions.
Drug testing and detection times
Hydrocodone, the active opioid in M365, is detectable on standard drug screenings. Detection windows vary by test type and individual factors (body mass, kidney function, frequency of use, hydration).
| Test type | Detection window | Notes |
|---|---|---|
| Urine | 2–4 days (single dose); up to 7 days (chronic use) | Most common workplace/clinical screen; may appear as “opiates” or specifically as hydrocodone on confirmatory GC/MS testing |
| Blood | 12–24 hours | Reflects current impairment; used in clinical emergencies |
| Saliva | 12–36 hours | Less common; used for roadside DUI testing in some states |
| Hair follicle | Up to 90 days | Reflects historical use pattern, not recent impairment |
Patients with a legitimate prescription should disclose it to the testing authority before the test. A prescription does not invalidate a positive result but may affect its interpretation in employment or legal contexts.
Seeking treatment for M365 pill misuse or addiction
If you or someone you care about is struggling with M365 pill misuse, prescription opioid dependence, or opioid use disorder, effective, evidence-based treatment is available. Recovery is not only possible — with the right support, it is the most common outcome.
Medication-assisted treatment (MAT)
The gold standard for opioid use disorder combines FDA-approved medications with behavioral therapy:
- Buprenorphine (Suboxone, Subutex). A partial opioid agonist that reduces cravings and withdrawal without producing significant euphoria at therapeutic doses. Available as an outpatient prescription.
- Methadone. A long-acting full opioid agonist dispensed through licensed opioid treatment programs (OTPs). Highly effective for severe OUD.
- Naltrexone (Vivitrol). An opioid antagonist that blocks opioid effects entirely. Available as a daily oral tablet or monthly injection. Requires a period of abstinence before initiation.
Levels of care
| Level | Description | Best for |
|---|---|---|
| Medical detox | 24/7 medically supervised withdrawal management, typically 3–7 days | Physical stabilization; anyone with health complications or severe withdrawal |
| Inpatient / residential | 24/7 structured therapeutic environment; 30–90+ days | Severe addiction, lack of safe home environment, co-occurring mental health disorders |
| PHP Indiana | Full-day programming, returns home evenings; ~5 days/week | Step-down from inpatient or intensive outpatient level of care |
| IOP Indiana | Group and individual therapy 3–5 days per week, 3 hrs/day | Stable living situation; maintaining work or school |
| Outpatient Rehab Indiana | Weekly therapy sessions; medication management appointments | Mild dependency; ongoing maintenance and relapse prevention |
For some people, misuse develops gradually. It may start with taking extra doses during severe pain, using the medication to sleep or relax, or feeling anxious about running out. When that pattern begins, drug rehab Indiana can help prevent the problem from becoming more severe.
✚ Get help now. Free confidential helpline. Call Red Ribbon Recovery Indiana at (317) 707-9848 (confidential, 24/7, 365 days/year). We provide drug rehab Indiana and treatment referrals for individuals facing substance use disorders and mental health issues.
Medical disclaimer. This article is intended for educational and informational purposes only. It does not constitute medical advice, diagnosis, or treatment recommendations. Always consult a qualified healthcare professional before starting, stopping, or changing any medication. In a medical emergency, call 911.
If you or someone you know is struggling with opioid use, call our helpline: (317) 707-9848 (24/7, confidential).
Sources
- U.S. Food and Drug Administration. (n.d.). Hydrocodone Bitartrate and Acetaminophen Tablets USP Full Prescribing Information. Mallinckrodt Pharmaceuticals.
- Drug Enforcement Administration, U.S. Department of Justice. (August 22, 2014). Schedules of Controlled Substances: Rescheduling of Hydrocodone Combination Products from Schedule III to Schedule II. Federal Register, 79 FR 49661.
- National Institute on Drug Abuse. (2024). Opioid Overdose Crisis.
- U.S. National Library of Medicine. (2024). Hydrocodone and Acetaminophen. StatPearls Publishing.
- Centers for Disease Control and Prevention. (2022). 2022 Clinical Practice Guideline for Prescribing Opioids for Pain. MMWR, 71(3), 1–95.
- Vowles, K. E., et al. (2015). Rates of opioid misuse, abuse, and addiction in chronic pain: a systematic review and data synthesis. Pain, 156(4), 569–576.
- Substance Abuse and Mental Health Services Administration. (2021). Medications for Opioid Use Disorder (TIP 63).
- U.S. National Library of Medicine. (n.d.). HYDROCODONE BITARTRATE AND ACETAMINOPHEN tablet — Mallinckrodt Inc. NDC 0406-0365. DailyMed.